How arousal differentially shapes sound processing in the brain.
By Ross Williamson, Ph.D.
Arousal—our fluctuating level of alertness—is a powerful modulator of brain function. It shapes how neurons respond to sensory input, yet most studies of arousal in the auditory brain have treated neurons as a single, homogeneous population.
In reality, the auditory cortex contains several distinct classes of excitatory neurons with different connectivity patterns and functional roles. Whether arousal modulates these cell types uniformly or in distinct ways has remained an open question.
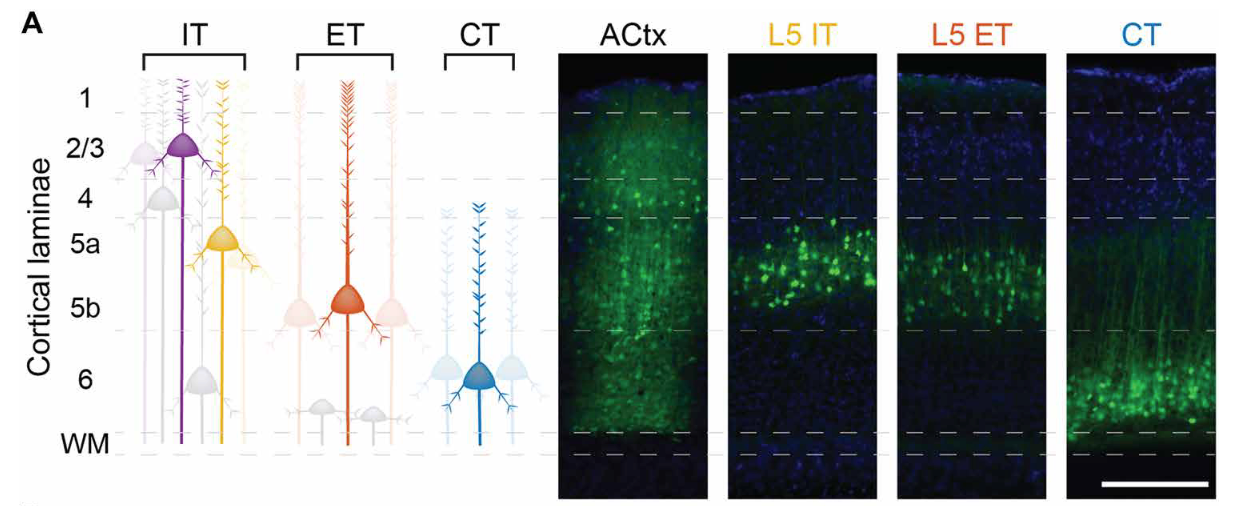
The study tracked arousal (via pupil size) and brain cell activity in the auditory cortex. Left: Illustrations of the different excitatory neuron types. Right: Brain images show active cells glowing green and all cell nuclei in blue. Credit: Kaufman, Krall, and Williamson/Science Advances
The auditory cortex sends information both within the cortex and to distant brain regions through specialized projection neurons. Among these, extratelencephalic (ET) neurons reside in the deep layers and project to subcortical structures including the midbrain, thalamus, striatum, and amygdala.
Corticothalamic (CT) neurons send feedback to the thalamus, while intratelencephalic (IT) neurons communicate within the cortex and with the contralateral hemisphere. These cell types differ not only in where they send information, but also in how they express receptors for the neuromodulatory systems that regulate arousal.
In our study, published in Science Advances in April 2026, graduate student Keith Kaufman, alongside postdoctoral researcher Rebecca Krall, Ph.D., used two-photon calcium imaging to record the activity of thousands of individual neurons across these excitatory subpopulations in the auditory cortex of awake mice.
Simultaneously, we tracked pupil diameter, a well-established physiological index of arousal, to determine how each cell type’s sound-evoked responses varied with the animal's internal state.
We found that arousal modulated each cell type differently. ET neurons exhibited a linear increase in response magnitude and stimulus encoding accuracy with rising arousal, driven by multiplicative and additive gain changes. CT and superficial layer neurons followed an inverted-U pattern, responding best at intermediate arousal levels. IT neurons were largely unaffected.
These differences in encoding accuracy closely tracked the stability, or reliability, of each population's neural responses, revealing a mechanistic link between internal state and representational fidelity.
These findings are relevant to understanding tinnitus. Previous work has demonstrated that ET neurons become hyperactive following noise-induced hearing damage, and tinnitus patients exhibit abnormally strong coupling between the auditory cortex and the very subcortical regions that ET neurons target: the amygdala and striatum.
By showing that arousal systematically modulates ET activity in the normal brain, our results identify a circuit-level mechanism through which stress and anxiety, both of which elevate arousal, could compound tinnitus-related hyperactivity in this descending pathway.
Understanding how internal state interacts with cell-type–specific circuits in the auditory cortex may ultimately help identify therapeutic targets for tinnitus and related hearing disorders.
Ross Williamson, Ph.D., is an assistant professor in the department of otolaryngology, neurobiology, and bioengineering at the University of Pittsburgh. He is a 2020 Emerging Research Grants recipient, which was renewed for a second year in 2022.
The team’s paper, “Pupil-linked arousal heterogeneously modulates cell-type–specific sensory processing,” was published in Science Advances in April 2026.







It took 20 years but Les Paul, who was born 111 years ago on June 9, 1915, forever changed music with his invention.