By Kishore Mosaliganti, Ph.D., and Ian Swinburne, Ph.D.
The inner ear senses sound for hearing and body movement for balance. In the embryo, the rudimentary ear grows from a fluid-filled balloon that is enveloped in a tight layer of cells. In our paper published in the journal eLife on Oct. 1, 2019, we examined how this balloon grows into the more complex ear. Our work helped us formulate a new mathematical theory on how ear growth in animals is controlled.
To do the research, we took advantage of the zebrafish embryo’s transparency by using a high-resolution microscope to take detailed 3D pictures of the inner ear as it grew during the first two days of fish development.
We observed that the ear grows dramatically and increases its volume by four times over a period of 24 hours. Most of the increase in size originates from the accumulation of fluid and not because of cell division to increase the tissue mass. This was a very surprising finding since most tissues in development grow by increasing the number or size of cells.
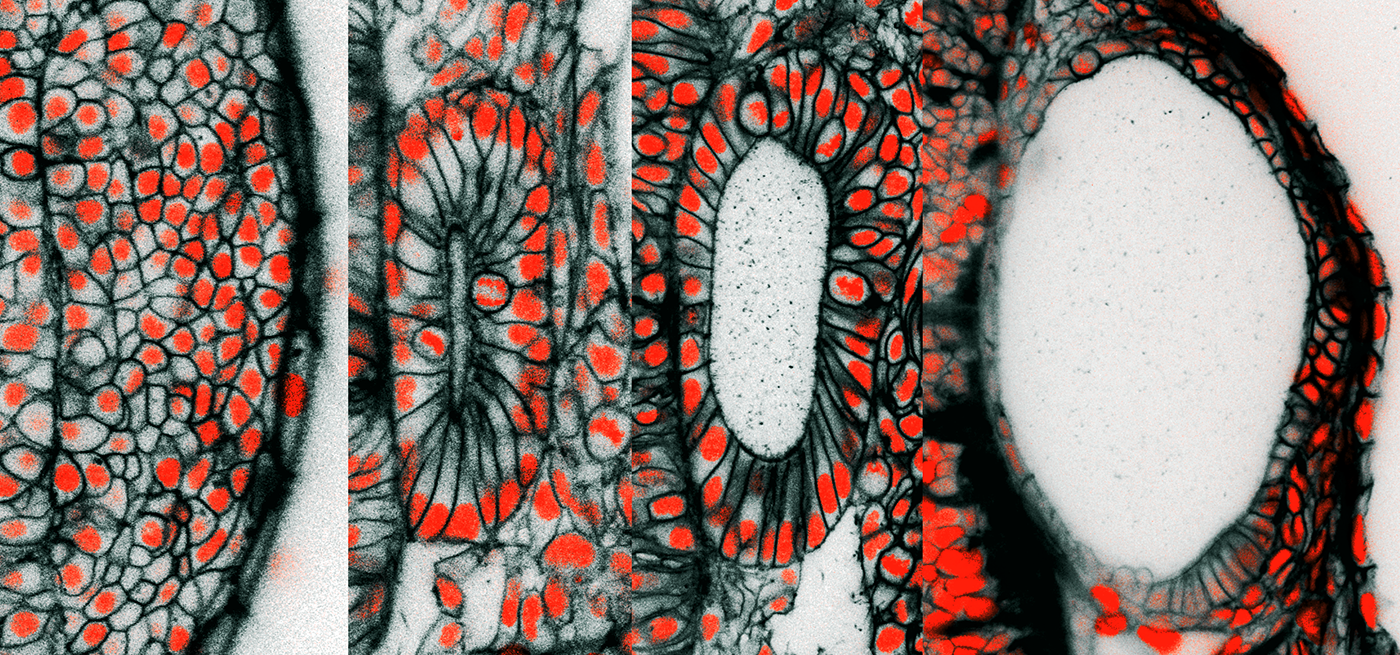
These are cross-sectional pictures from live 3D microscopic images of the developing zebrafish ear. The images are from time-points at 12, 16, 24, and 45 hours after egg fertilization, a span of time during which the ear rudiment grows more than four times in volume. The black coloring is a fluorescent protein targeted to the outer membrane of individual cells. The red is from a fluorescent protein targeted to the nucleus of individual cells where it binds to DNA. At 45 hours post-fertilization, the length of the embryonic ear’s long axis is approximately 1/10th the thickness of a penny.
A second observation was that the ear pushed out the neighboring brain structures, and that the ear cells that envelop the balloon appear stretched.
Both of these observations suggested to us that pressure within the developing ear was increasing. We wondered if cells control this pressure directly or if it is just a byproduct of the growth process. We decided to dive deeper.
We designed and built a nanoscale pressure probe small enough to insert into the tiny ear of the fish embryo and sensitive enough to detect the first increase in fluid pressure. To examine if the pressure is monitored and controlled, we popped the ear with needles and we observed that the ear would collapse, much like a balloon that has been popped. To our amazement, we watched the popped embryonic ear recover and rapidly catch up in size to the unpopped ear by briefly accelerating its inflation with fluid.
Just as an engineer may design a thermostat to maintain the temperature of a room, we began to think that the developing ear has a way to tell itself when it has reached the correct size by monitoring the internal fluid pressure. We developed a mathematical model of this process.
Next, we began to use this model to predict and test if the pressure also shapes the ear. The mechanical properties of the enveloping cells offer resistance to growth much like a waist belt. If the belt’s elasticity is locally changed, then growth rates can be controlled locally. This helps explain how the inner ear changes from a spherical shell to an elongated football-like shape.
To summarize, our paper demonstrates how biology does not limit itself to gene regulation and protein activities. Mechanics matter. To function in a 3D world where there is inertia and resistance to growth, an embryo and its developing organs actually control physical forces and collect feedback on these forces to inform regulatory processes. This is yet another reason why animal development is so orchestrated, robust, and precise.
Additionally, the crosstalk between mechanical forces and the behavior of cells in the ear is important to understand when investigating hearing and balance diseases where inner ear fluid pressure is out of control, such as Pendred syndrome and Ménière’s disease.
Ian Swinburne, Ph.D., is a postdoctoral researcher in the department of systems biology at Harvard Medical School, Massachusetts, where his colleagues included Kishore Mosaliganti, Ph.D. Swinburne is a 2019, 2018, and 2013 Emerging Research Grants recipient.
This groundbreaking research is made possible by generous individuals like you. Be part of our mission to prevent, treat, and cure hearing and balance conditions.